Laparoscopic Sleeve Gastrectomy – A Newcomer to Bariatric Surgery

By Stacy Brethauer, MD and Philip Schauer, MD
Fall 2007
As patients begin investigating surgical weight-loss options, one of the first questions to arise is “what procedure is best for me?” The answer to this question must come after thorough research regarding the risk and benefits of each procedure and an evaluation to determine the individual patient’s risk for undergoing surgery.
About 80 percent of the bariatric procedures performed in the United States are gastric bypass procedures. The other 20 percent are comprised of restrictive procedures, such as the laparoscopic adjustable gastric band. The laparoscopic sleeve gastrectomy (LSG), a relative newcomer to bariatric surgery, is growing in popularity.
The sleeve gastrectomy originated as the restrictive part of the duodenal switch operation. In the last several years, though, it has been used by some surgeons as a staging procedure prior to a gastric bypass or duodenal switch in very high-risk patients. It has also been used as a primary, stand-alone procedure by some surgeons.
How is Sleeve Gastrectomy Performed?
The majority of sleeve gastrectomies performed today are completed laparoscopically. This involves making five or six small incisions in the abdomen and performing the procedure using a video camera (laparoscope) and long instruments that are placed through these small incisions.
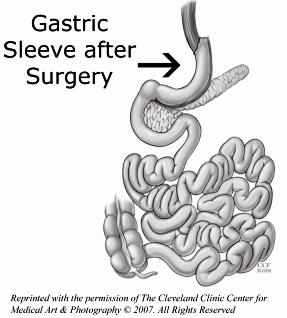
During the sleeve gastrectomy, about 75 percent of the stomach is removed leaving a narrowgastric tube or “sleeve” (pictured right). No intestines are removed or bypassed during the sleeve gastrectomy. This procedure takes one to two hours to complete. This short operative time is an important advantage for patients with severe heart or lung disease.
How Does the Sleeve Gastrectomy Cause Weight-Loss?
Sleeve gastrectomy is a restrictive procedure. It greatly reduces the size of the stomach and limits the amount of food that can be eaten at one time. It does not cause decreased absorption of nutrients or bypass the intestines. After this surgery, patients feel full after eating very small amounts of food. Sleeve gastrectomy may also cause a decrease in appetite. In addition to reducing the size of the stomach, the procedure reduces the amount of the “hunger hormone,” ghrelin, produced by the stomach.1 The duration of this effect is not clear yet, but most patients have significantly decreased hunger after the operation.
Who Should Have a Sleeve Gastrectomy?
This operation has been used successfully for many different types of bariatric patients.2 Since it is a relatively new procedure, there is no data regarding weight-loss, complications or weight regain beyond three years. At the Cleveland Clinic, we use this procedure as part of a staged approach for high-risk patients. Patients who have a very high body mass index (BMI) or severe heart or lung disease may benefit from a shorter, lower risk operation such as the sleeve gastrectomy as a first stage procedure. Sometimes, the decision to proceed with the sleeve gastrectomy is made in the operating room due to an excessively large liver or extensive scar tissue to the intestines that make gastric bypass impossible.
In patients who undergo LSG as a first stage procedure, the second stage (gastric bypass) is performed 12 to 18 months later after significant weight-loss has occurred, the liver has decreased in size and the risk of anesthesia is much lower. Though this approach involves two procedures, we believe it a safe and effective strategy for selected high-risk patients.
LSG is also being used as a primary weight-loss procedure in lower BMI patients. Because this is a more recent application of this procedure, it is currently being performed as part of an investigational protocol for this lower BMI patient group.
How Much Weight-loss Occurs after LSG?
Several studies have documented excellent weight-loss up to three years after LSG.3-6 In higher BMI patients who undergo LSG as a first stage procedure, the average patient will lose 40 – 50 percent of their excess weight in the first two years after the procedure.4 This typically equates to about 125 pounds of weight-loss for patients with a BMI greater than 60.
Patients with lower BMI’s who undergo LSG will lose a larger proportion of their excess weight (60 – 80 percent) within three years of the surgery. 3, 5, 6 Weight-loss after LSG has been directly compared to Laparoscopic Adjustable Gastric Banding (LAGB). In a randomized trial comparing LSG to LAGB, LSG resulted in better weight-loss at three years (66 percent versus 48 percent excess weight-loss). Additionally, more than 75 percent of patients will have significant improvement or resolution of major obesity-related co-morbidities such as diabetes, hypertension, sleep apnea and hyperlipidemia following sleeve gastrectomy.4, 5, 7, 8
What are the Risks of Sleeve Gastrectomy?
The risk of major postoperative complications after LSG is 5-10 percent, which is less than the risk associated with gastric bypass or malabsorptive procedures such as duodenal switch. This is primarily because the small intestine is not divided and reconnected during LSG as it is during the bypass procedures. This lower risk and shorter operative time is the main reason we use it as a staging procedure for high-risk patients.
Complications that can occur after LSG include a leak from the sleeve resulting in an infection or abscess, deep venous thrombosis or pulmonary embolism, narrowing of the sleeve (stricture) requiring endoscopic dilation and bleeding. Major complications requiring re-operation are uncommon after sleeve gastrectomy and occur in less than 5 percent of patients.
Is LSG a Good Choice for Me?
You should first know the risks and benefits of sleeve gastrectomy, adjustable gastric banding and gastric bypass. For high-risk patients and patients with very high BMI’s, we discuss LSG as a first-stage procedure prior to gastric bypass. We are also conducting a clinical trial that includes sleeve gastrectomy for lower BMI patients with diabetes. Ultimately, the decision regarding which procedure to perform is based on each patient’s operative risk and their expectations and goals for surgical weight-loss.
Sleeve Gastrectomy May be Performed for the Following Reasons:
|
About the Authors:
Stacy Brethauer, MD, is a staff surgeon at the Cleveland Clinic specializing in advanced laparoscopy and bariatric surgery. He is an active member of the American Society for Metabolic & Bariatric Surgery and is co-editor of Minimally Invasive Bariatric Surgery. He has a special interest in endoscopic procedures and emerging technologies that benefit bariatric patients.
Philip Schauer, MD, is past president of the American Society for Metabolic & Bariatric Surgery and is the Director of the Bariatric and Metabolic Institute at the Cleveland Clinic. He has been published extensively on bariatric procedures and outcomes and has been instrumental in promoting the field of bariatric surgery worldwide through his many leadership roles.
References:
1. Langer FB, Reza Hoda MA, Bohdjalian A, et al. Sleeve gastrectomy and gastric banding: effects on plasma ghrelin levels. Obes Surg 2005; 15(7):1024-9.
2. Baltasar A, Serra C, Perez N, et al. Laparoscopic sleeve gastrectomy: a multi-purpose bariatric operation. Obes Surg 2005; 15(8):1124-8.
3. Lee CM, Cirangle PT, Jossart GH. Vertical gastrectomy for morbid obesity in 216 patients: report of two-year results. Surg Endosc 2007.
4. Cottam D, Qureshi FG, Mattar SG, et al. Laparoscopic sleeve gastrectomy as an initial weight-loss procedure for high-risk patients with morbid obesity. Surg Endosc 2006; 20(6):859-63.
5. Moon Han S, Kim WW, Oh JH. Results of laparoscopic sleeve gastrectomy (LSG) at 1 year in morbidly obese Korean patients. Obes Surg 2005; 15(10):1469-75.
6. Himpens J, Dapri G, Cadiere GB. A prospective randomized study between laparoscopic gastric banding and laparoscopic isolated sleeve gastrectomy: results after 1 and 3 years. Obes Surg 2006; 16(11):1450-6.
7. Hamoui N, Anthone GJ, Kaufman HS, Crookes PF. Sleeve gastrectomy in the high-risk patient. Obes Surg 2006; 16(11):1445-9.
8. Silecchia G, Boru C, Pecchia A, et al. Effectiveness of laparoscopic sleeve gastrectomy (first stage of biliopancreatic diversion with duodenal switch) on co-morbidities in super-obese high-risk patients. Obes Surg 2006; 16(9):1138-44.
by OAC Staff Members Kendall Griffey and Michelle “Shelly” Vicari Winter 2024 In a world that often…
Read Articleby Rachel Engelhart, RD; Kelly Donahue, PhD; and Renu Mansukhani, MD Summer 2023 Welcome to the first…
Read ArticlePost-operative addiction is often overly simplified as transfer addiction or cross-addiction, assuming individuals “trade” compulsive eating for…
View Video









